Choosing the right battery is important for every device or car. A battery cell chemistries comparison is key to making an informed decision. Each chemistry has its own advantages and disadvantages, including energy density, performance, and cost. Lithium-ion batteries are very popular, comprising 62.4% of the world market, which indicates their widespread use in new technology. The table below illustrates how LiFePO4 and NMC differ in terms of energy density and suitability for various applications:
Battery Chemistry | Energy Density | Performance | Application Suitability |
|---|---|---|---|
LiFePO4 (LFP) | Lower | Good | Cost-sensitive, EVs, energy storage |
NMC | Higher | Excellent | High-performance EVs, long-range applications |
Selecting the right battery features based on a battery cell chemistries comparison helps you achieve the best results for your needs.
Key Takeaways
Picking the right battery chemistry is very important for how well things work and how much they cost. Lithium-ion batteries are used the most because they store a lot of energy and last a long time. This makes them great for electric cars and small gadgets. Safety matters a lot too. Lithium iron phosphate (LiFePO4) batteries are some of the safest for storing energy at home. Knowing about energy density and cycle life helps you choose the best battery for things like electronics or big power storage. Recycling batteries is very important to help the environment and get back useful materials, so always think about recycling.
Battery Cell Chemistries Comparison

Key Metrics Overview
A battery cell chemistries comparison helps people pick the best battery. Many batteries are used in things like cars, phones, and big energy systems. Each chemistry has good and bad points. To compare them, we look at important things.
Chemistry | Cell Voltage (V) | Energy Density (MJ/kg) | Self-Discharge (%/month) | Cycle Life (max) |
|---|---|---|---|---|
NiCd | 1.2 | >0.14 | 20 | 800 |
Lead Acid | 2.2 | >0.14 | 15 | 300 |
NiMH | 1.2 | >0.36 | 30 | 500 |
Lithium ion | 3.6 | >0.46 | 10 | 1000 |
Lithium Cobalt Oxide | 3.6 | >0.72 | 5 | 500 |
Lithium Iron Phosphate | 3.3 | >0.32 | 5 | 12000 |
Lithium Nickel Manganese Cobalt Oxide | 3.7 | >0.54 | 5 | 1000 |
Lithium Titanate | 2.4 | >0.23 | 5 | 20000 |
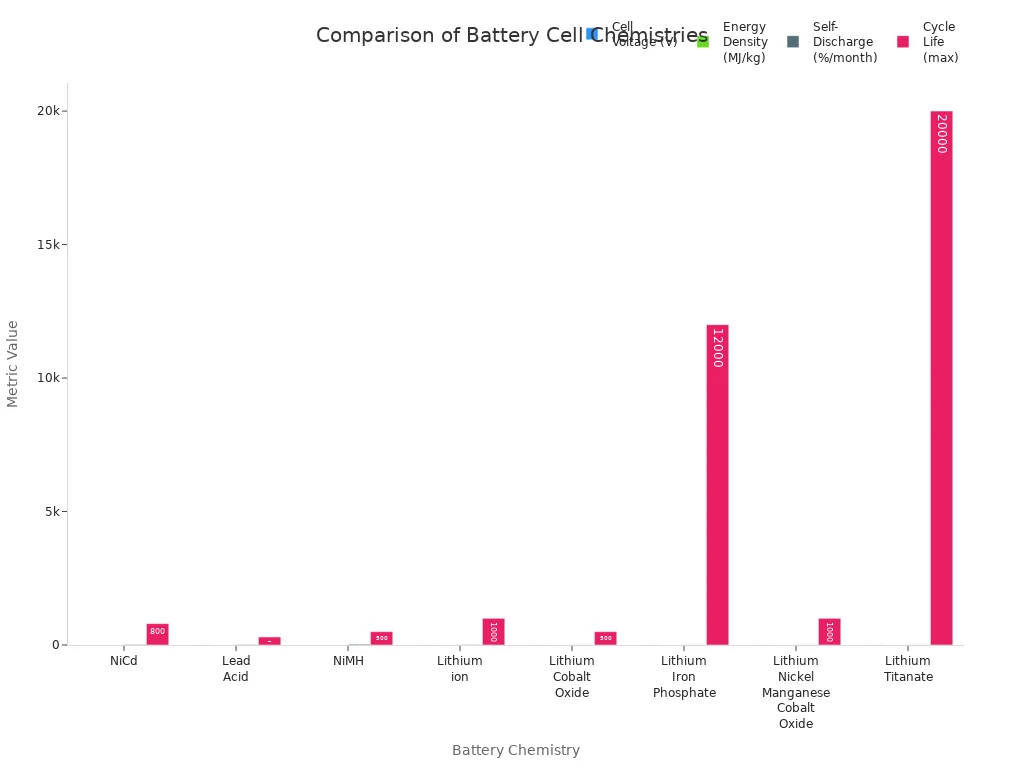
These numbers show how each battery works in real life. Cell voltage means how much power the battery gives. Energy density tells how much energy it holds for its weight. Self-discharge shows how fast a battery loses power when not used. Cycle life is how many times you can use and charge the battery before it stops working.
Application Relevance
Comparing battery cell chemistries gets harder when you think about how they are made and used. How a battery is made changes its shape, size, and how well it works. Cylindrical batteries are tough and last a long time, so they are good for power tools. Prismatic batteries are better for small spaces, so they fit in phones and laptops. Pouch cells are light and bendy, so they work in odd-shaped devices.
No one battery chemistry is perfect for everything. Each use, like cars or big energy storage, needs a balance of price, weight, safety, and how well it works.
The most common battery cell chemistries in today’s technology are:
Lithium-ion: Found in most small electronics and electric cars. It is also used in almost all grid energy storage.
Sodium-Ion: A cheap choice for grid storage and some cars.
Lithium-Sulfur: Light and stores lots of energy, but does not last long.
Lithium-Metal: Can help electric cars go farther on one charge.
Flow Batteries: Give steady power for a long time in grid storage.
Vanadium-Redox Flow: Stores energy from things like solar and wind.
Zinc-Polyiodide Flow: Holds more energy than other flow batteries.
Sodium-Metal Halide: Used for grid storage that does not move.
Zinc-air: Makes power by using air.
Zinc-Manganese Oxide: Uses cheap stuff and stores more energy than lead-acid.
Lead-acid: Trusted and low-cost for some jobs.
A battery cell chemistries comparison should look at all these things. The best battery depends on what it will power and what the user needs. Some batteries last longer, some are safer, and some are cheaper. Makers must pick the right chemistry for the job to get the best results.
Energy Density Comparison

Volumetric Energy Density
Volumetric energy density tells us how much energy fits in a space. This is important for things that need to be small or light, like phones or electric cars. If a battery has higher volumetric energy density, it can store more power in less space.
The table below shows how much energy different batteries can hold in a certain space:
Energy Density (Wh/kg) | |
|---|---|
Lead acid | 30-50 |
Nickel-cadmium | 45-80 |
Nickel-metal hydride | 60-120 |
Lithium-ion | 50-260 |
Lithium-ion batteries can hold up to 260 Wh/kg. Nickel-metal hydride batteries are also good, but lead acid batteries hold the least. This comparison helps engineers pick the best battery for small devices.
Tip: Laptops and electric cars often use lithium-ion batteries. They give lots of energy and do not take up much space.
Gravimetric Energy Density
Gravimetric energy density shows how much energy a battery has for its weight. This is important for things that move, like electric cars, drones, or small electronics. Lighter batteries with high gravimetric energy density help these things work longer without getting heavy.
Here is a table that shows how much energy different batteries have for their weight:
Energy Density (Wh/kg) | |
|---|---|
Lithium-ion | 0.46 – 0.72 |
Nickel-Cadmium (NiCd) | 0.14 – 1.08 |
Nickel-Metal Hydride (NiMH) | 0.4 – 1.55 |
Lead-Acid | N/A |
Lithium-ion batteries do very well here. Nickel-metal hydride batteries can also have high numbers, but lead-acid batteries do not do as well. When engineers need batteries for things that must be light, gravimetric energy density is very important.
Note: If a battery has higher gravimetric energy density, portable devices can run longer.
Battery Cell Comparison: Specifications
Cycle Life & Charge Time
Cycle life means how many times you can use a battery. It is the number of times you can charge and use it before it gets weak. Charge time is how fast a battery fills up with energy. These things are important for things that need to last long or charge quickly.
The table below shows how long some batteries last:
Battery Chemistry | |
|---|---|
LiFePO4 | 2,000 to 10,000 cycles |
NMC | 1,000 to 2,500 cycles |
LTO | 10,000 to 20,000 cycles |
LiFePO4 batteries work longer than NMC batteries. LTO batteries last the longest and are good for heavy use. Most lithium-ion batteries charge faster than old types. Fast charging is helpful for electric cars and small gadgets.
Internal resistance changes how fast a battery charges. If resistance is low, the battery charges and works faster. The table below shows resistance for some batteries:
Battery Chemistry | |
|---|---|
Nickel-Cadmium | 155 |
Nickel-Metal-Hydride | 778 |
Lithium-Ion | 320 |
Nickel-cadmium batteries have less resistance than nickel-metal hydride. Lithium-ion batteries have a good mix of resistance and power.
Safety & Maintenance
Safety is very important when picking a battery. Some batteries can get too hot or even catch fire. Others might leak bad chemicals. The table below shows some risks and how to stay safe:
Mitigation Measures | ||
|---|---|---|
Lithium-ion | Thermal runaway, fire risk | Battery management systems, thermal cutoffs |
Lead-acid | Hydrogen gas release, acid spills | Ventilation, sealed batteries, safe handling |
Sodium-ion | Overheating | Thermal management systems |
Lithium-ion batteries can burn if they get too hot or break. Special systems help keep them safe. Lead-acid batteries can let out gas or spill acid. They need good air flow and careful use. Sodium-ion batteries can get hot, but better controls help stop problems.
Different batteries need different care. The table below shows what each type needs:
Battery Type | |
|---|---|
Lithium-ion | Keep charge between 20-80%, avoid full discharge and overcharging, charge safely. |
Lead-acid | Check electrolyte levels, charge properly to avoid sulfation, limited cycle life. |
Nickel-cadmium | Full discharge sometimes to prevent memory effect, regular charging. |
Nickel-metal hydride | Regular charging, avoid deep discharges, less maintenance than lead-acid. |
Lithium-ion batteries need safe charging but not much else. Lead-acid batteries need checks and the right charging. Nickel-cadmium batteries need to be used up sometimes to avoid memory problems. Nickel-metal hydride batteries need less care but still need charging often.
Environmental Impact
Batteries can hurt the environment in many ways. Making and throwing away batteries can cause pollution. Some batteries use metals that are hard to get or recycle. Others have dangerous chemicals.
Lithium-ion batteries need lithium from the ground, which can hurt nature. Recycling helps lower the harm.
Lead-acid batteries have lead and acid, which are bad if not handled right. Recycling keeps these out of nature.
Nickel-cadmium batteries have cadmium, which is very toxic. Special recycling keeps cadmium out of air and water.
Nickel-metal hydride batteries are safer than nickel-cadmium but still need careful recycling to get the metals back.
Recycling batteries saves energy and helps stop pollution. Safe recycling and disposal protect people and the earth.
A battery cell comparison should always think about the environment. Picking batteries that last longer and are easy to recycle helps the planet.
Lithium-Ion Battery and Other Chemistries
Li-ion Variants
Lithium-ion battery technology has many types. Each type is good for different things. The most common types are lithium iron phosphate (LiFePO4), lithium nickel manganese cobalt oxide (NMC), and lithium manganese oxide (LMO). These batteries are not the same in voltage, energy, or how long they last.
Battery Type | Voltage | Specific Energy | Cycle Life | Applications |
|---|---|---|---|---|
Lithium Iron Phosphate (LiFePO4) | 3.20V | 90–120Wh/kg | 2000+ cycles | Energy storage, portable applications |
Lithium Nickel Manganese Cobalt (NMC) | 3.6–3.7V | 160–270Wh/kg | 1000–2000 cycles | Electric vehicles, medical devices |
Lithium Manganese Oxide (LMO) | 3.7V | 120–170Wh/kg | N/A | Power tools, medical devices, security systems |
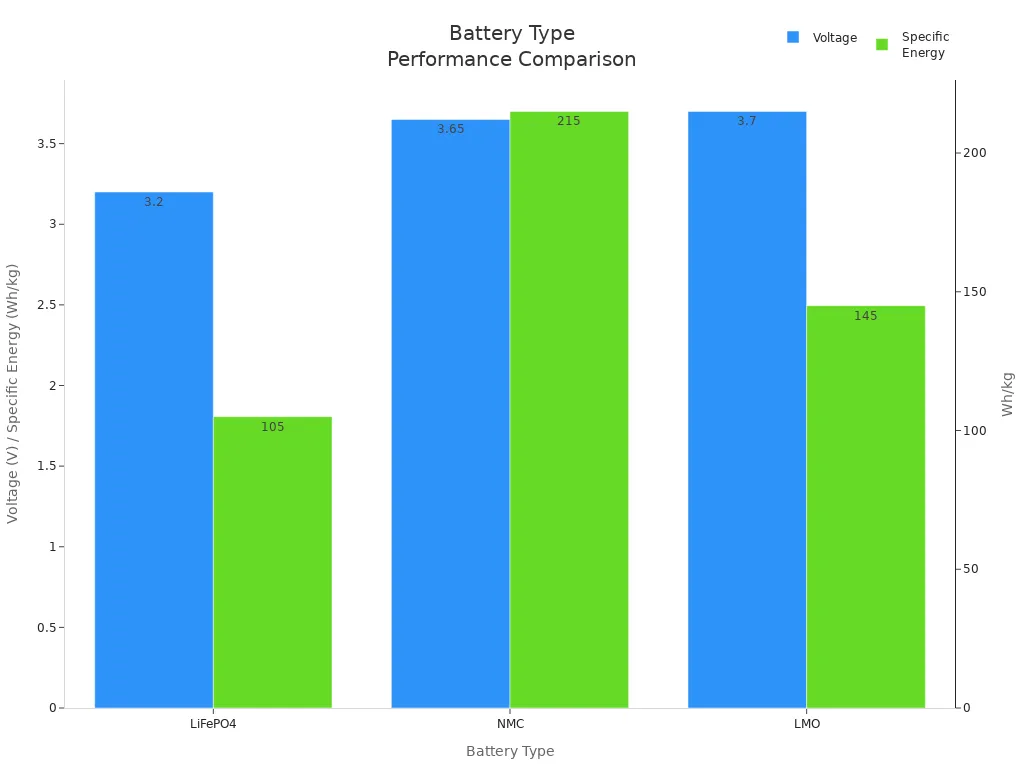
NMC batteries can hold the most energy. They work well in electric cars. LiFePO4 batteries last longer and are safer. They are good for storing energy. LMO batteries give strong power fast. They are used in power tools and security systems.
Tip: Each lithium-ion battery type is good at something. Pick the one that matches your needs.
Lead Acid, NiCd, NiMH
Older battery types like lead acid, nickel-cadmium, and nickel-metal hydride have been used for a long time. Each one has good and bad points.
Battery Type | Advantages | Disadvantages |
|---|---|---|
Lead Acid | High current output, low upfront cost | Large, heavy, slow charging, shorter lifespan, not environmentally friendly |
Nickel-Cadmium | Higher energy density, faster charge time, longer cycle life | Memory effect, high self-discharge, heavy, contains toxic cadmium |
High energy density, high cycle life, low self-discharge, low maintenance | Requires protection circuit, potential fire risk, higher cost, recycling challenges |
Lead acid batteries are cheap and give strong power. But they are heavy and do not last long.
Nickel-cadmium batteries charge fast and last longer. But they can lose power if not used right and have harmful cadmium.
Nickel-metal hydride batteries are safer and store more energy than nickel-cadmium. But they are still heavier than lithium-ion batteries.
Lithium-ion batteries stand out because they store lots of energy, last a long time, and need little care. But they must be handled safely and cost more to make. Each battery type is best for certain jobs. Engineers choose the right one for what the device needs.
Matching Chemistries to Applications
Electric Vehicles
Electric vehicles need batteries that hold lots of energy and last long. Two main chemistries are used most:
Lithium Iron Phosphate (LFP): This type is very safe and lasts many cycles. It works well in electric buses and cheaper cars.
Lithium Nickel Manganese Cobalt Oxide (NMC): This one stores more energy, so it is good for cars that go far.
Energy density is very important for electric vehicles. If a battery has more energy density, the car can drive farther before charging. Most electric cars today use lithium-ion batteries with energy densities from 150 to 250 Wh/kg. This lets many cars travel 200 to 400 miles before they need to charge again.
Energy Density | Operating Temperature Range | Size Requirement | |
|---|---|---|---|
Lithium-ion (Li-Ion) | High | Up to 60°C | Smaller |
Lithium Iron Phosphate (LFP) | Lower | Below 0°C | Larger |
Tip: NMC batteries are best for long trips. LFP batteries are safer and good for city driving.
Consumer Electronics
Phones, laptops, and tablets need batteries that are light and strong. Lithium-ion and lithium-polymer batteries are used the most. They have high energy density, last long, and do not lose much charge when not used.
Battery Chemistry | Charge Density | Discharge Rate | Cost | Preferred Use |
|---|---|---|---|---|
Lithium-ion | High | Moderate-High | Moderate | Rechargeable devices |
Lithium-polymer | Very High | High | High | High-performance devices |
NiMH | Moderate | Moderate | Low | Older devices |
Most gadgets use lithium-ion batteries.
High-end phones and drones use lithium-polymer batteries.
Older electronics use nickel-metal hydride batteries.
Note: Lithium-ion batteries are lighter and safer than old types. They also do not have memory effect.
Grid Storage
Grid storage helps balance energy from solar and wind. These systems need batteries that last many years and can be charged and used many times.
Battery Type | Advantages | Limitations |
|---|---|---|
Lithium-Ion | High energy density, long cycle life | Limited lifespan compared to some alternatives |
Flow Batteries | Scalable, long cycle life, quick response | Lower power density, complex management |
Sodium-Sulfur | High energy density, efficient for large-scale use | Needs high temperatures, careful management |
Cycle life is very important for grid storage. Lithium iron phosphate batteries can last 3,000 to 10,000 cycles. Flow batteries last even longer and can be made bigger for large projects.
Industrial Uses
Industrial machines need batteries that are tough and work well. These batteries must handle heat, shaking, and heavy use.
Battery Chemistry | Key Features | Suitable Applications |
|---|---|---|
Lithium-ion (Li-ion) | High energy, long life | Portable tools, vehicles |
Lead-acid | Robust, low cost | Backup power, forklifts |
Nickel-metal hydride | Good safety, moderate energy | Hybrid vehicles, equipment |
Sodium-ion | Cost-effective, sustainable | Large-scale energy storage |
Flow batteries | Long cycle life, scalable | Grid-scale storage |
Lithium batteries give great performance and need little care for most industrial jobs.
When picking a battery, think about energy, safety, price, and how long it will last. Each job has a battery that fits it best.
No one battery chemistry works for everything. You have to pick based on what you need. Think about energy density, power density, cycle life, safety, and what you will use it for.
Key Aspect | Description |
|---|---|
Energy Density | How much energy fits in a certain space. |
Power Density | How fast the battery can give out energy. |
Cycle Life | How many times you can use and charge it before it gets weak. |
Safety | How likely it is to fail or be dangerous. |
Application Focus | If it works well for electronics, cars, or big energy storage. |
To find the right battery, you should check if you can recharge it. You also need to think about how much space and weight you have. Look at how much voltage and power you need. Make sure the battery will last long enough for your use.
There are lots of websites and articles that help you compare batteries. These can show you the good and bad sides for each job.
FAQ
What is the safest battery chemistry for home use?
Lithium iron phosphate (LiFePO4) batteries are very safe. They do not get too hot easily. They almost never catch fire. Many people use them for storing energy at home.
Why do electric cars use lithium-ion batteries?
Electric cars use lithium-ion batteries because they hold lots of energy in a small space. These batteries last longer than old types. They also weigh less than other batteries.
Can batteries be recycled?
Most batteries can be recycled. Recycling gets back useful metals. It also helps stop pollution. Many stores and recycling places take old batteries.
Which battery lasts the longest?
Lithium titanate (LTO) batteries last the longest. They can be charged up to 20,000 times. These batteries are good for things that need to work a long time.



